What are the types of combination reaction? The law of multiple proportions states that, when two elements combine to form more than one compound, the mass of one element, which combines with a fixed mass of the other element, will always bear a simple ratio to the other. Can two elements form more than one compound? Water molecules are made of hydrogen and oxygen atoms, in the definite proportion of two hydrogens for one oxygen. Water is a compound because it is made up of water molecules. So, the new element would probably belong to the noble gas group. There are few known noble gas compounds, such as xenon and krypton compounds. What elements can combine?Įxplanation: The unreactive elements on the periodic table are the noble gases, and the reason for that is because they usually have octet shells, hence their unreactivity. Six of these occur in extreme trace quantities: technetium, atomic number 43 promethium, number 61 astatine, number 85 francium, number 87 neptunium, number 93 and plutonium, number 94. Of these 118 elements, 94 occur naturally on Earth. 
How many elements do we have all together? By alloying, some of the important properties of metals can be improved. What are alloys? Mixtures of metals, called alloys, are more commonly used than the pure metal. Electrons are located in energy levels that occur at certain distances from the nucleus, called shells. These bonds involve the electrons that orbit the nucleus of the atom. The answer is that compounds are formed when elements are joined and held together by strong forces called chemical bonds. The conservation of mass laws, the law of definite proportion, and the law of multiple proportions. One example of a combination reaction of two elements is Ba (s) + F2 (g) –> BaF2 (s). 
What is the formula of combination reaction?Ī combination of one element and one compound reaction occurs when an element and a compound react to form one compound. 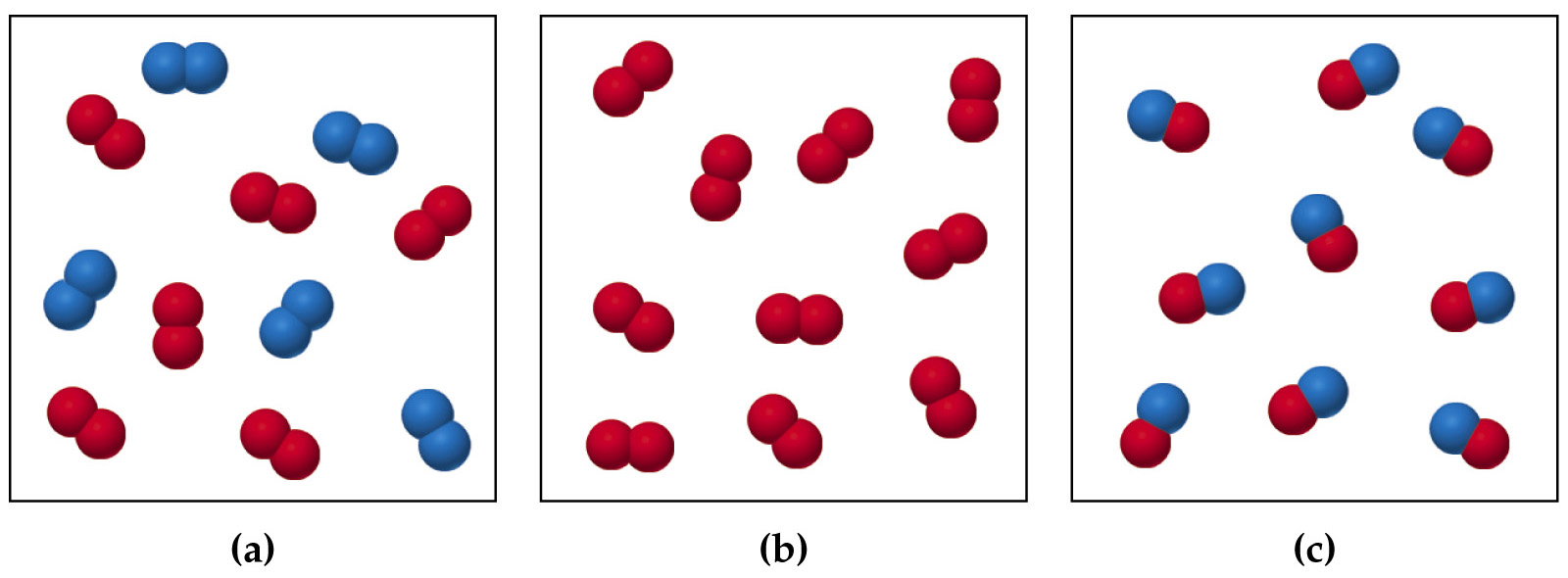
Simplification of the overall equation will eliminate chemical species that exist on both sides of the equation without change. You combine multiple reactions into a single equation by listing all the reactants on the left side of the equation and all the products on the right side of the equation. Why do atoms combine in elements?Ītoms combine together mainly for two reasons Firstly, they combine together to form various compounds. If “touching” is taken to mean that two atoms influence each other significantly, then atoms do indeed touch, but only when they get close enough. When elements combine or join together new substances are formed. How do you turn an element into a compound? Can all elements react with each other?Īll elements, apart from the noble gases, are chemically reactive, because they are more stable electronically when they share electrons with other atoms. If it’s literally the entire periodic table, the immediate decay of unstable elements will cause an immense explosion and radioactive cloud. What happens if we combine all the elements?īasically, the entire mass will violently explode spaying the area with toxic and radioactive material. The atoms of molecules are linked together through a reaction known as chemical bonding. Note:Atoms, in a sense, are tied together to form molecules. The atoms share their valence electrons and bond together, forming a molecule. One of the simplest is called methane, in which there are always four times as many hydrogen particles as carbon particles. The elements carbon and hydrogen combine to form many different compounds. A single neutral atom has a number of electrons surrounding its nucleus that is the same as the number of protons in the nucleus of the atom.Ī compound is a substance that contains two or more elements chemically combined in a fixed proportion. Ionic Bonds Another way in which atoms can be joined together is by transfer of electrons from one atom to another. So, if you were to combine one atom of each of the elements from the periodic table it would leave only a TINY group of compounds and the rest would still be sitting around as elements. Non-reactive elements like helium, neon, argon, krypton, xenon and radon ignore all other elements and remain unchanged. Compounds are commonly termed as chemical compounds. What is combining two elements called?Īccording to definition of compounds, two or more elements combine together chemical to form a new substance called compound. Compounds may be formed by combining atoms of their constituent elements by ionic bonds or by covalent bonds. 24 Is human made of atoms? What is the process of combining elements?Įlements can be chemically combined into compounds, therefore, a compound consists of two or more elements combined, in definite proportions, by chemical means. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
